Synthesis, optical properties and cation mediated tuning of the reduction potentials of core-annulated naphthalene diimide derivatives†
Abstract
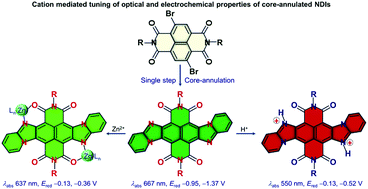
Napthalene diimides (NDIs) are attractive candidates for electrical energy storage owing to the stabilisation of complexes between electrogenerated dianions and cations. However, the stability of such complexes is often compromised due to the monodentate nature of the electrogenerated carbonyl anions. To overcome the limitations, herein we synthesised two new core-annulated NDIs, in which additional coordinating groups have been installed adjacent to the carbonyl groups, converting the dyes into multidentate ligands. The new NDIs, aImPyDI and bImPyDI, exhibited a significant change in their reduction potentials in the presence of Zn2+ ions. The imides also displayed similar reduction potentials which can be tuned in the presence of protons. Additionally, the dyes revealed rare negative-halochromism in the presence of an acid. Both the dyes showed a good emission quantum yield.


 Please wait while we load your content...
Please wait while we load your content...