Platinum on carbon-catalysed site-selective H–D exchange reaction of allylic alcohols using alkyl amines as a hydrogen source†
Abstract
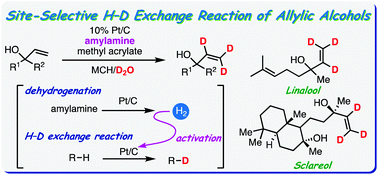
We developed a platinum on carbon-catalysed deuteration reaction of tert-allylic alcohols using deuterium oxide as a deuterium source. Amylamine was dehydrogenated by platinum on carbon to generate an appropriate amount of hydrogen gas, which efficiently promoted the H–D exchange reaction. Various allylic alcohols were site-selectively deuterated with good to excellent deuterium contents without hydrogenation of the alkene functionality. The obtained deuterium-labelled allylic alcohols were further transformed into various deuterium-labelled building blocks without degradation of the original deuterium content.


 Please wait while we load your content...
Please wait while we load your content...