Graphene in situ composite metal phthalocyanines (TN-MPc@GN, M = Fe, Co, Ni) with improved performance as anode materials for lithium ion batteries
Abstract
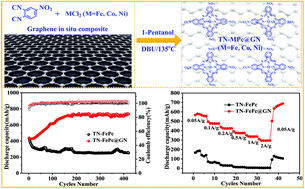
In view of the disadvantage of the limited active site utilization due to the easy aggregation of phthalocyanine compounds, three kinds of graphene composite metal phthalocyanines (TN-MPc@GN, M = Fe, Co, Ni) were prepared using an in situ composite method, and their electrochemical properties were investigated as anode materials for lithium-ion batteries. The layered structure and large specific surface area of graphene are used to disperse phthalocyanine uniformly, so that the conjugated structure (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/C
C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N/N
N/N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of phthalocyanine is more exposed and the utilization of active sites for embedding lithium ions are greatly enhanced. At the same time, the large surface area is beneficial to the contact between materials and the electrolyte, which results in a faster transmission path. As a result, the first discharge specific capacities of the graphene composite phthalocyanine (TN-MPc@GN, M = Fe, Co, Ni) electrodes are 440.8, 765.0 and 753.9 mA h g−1, respectively. After 420 cycles, the specific capacities increased to 734.6, 844.7 and 949.1 mA h g−1, respectively, which are much higher than those of the pristine three phthalocyanine (TN-MPC, M = Fe, Co, Ni) electrodes without the graphene composite. Using the graphene composite can be an effective strategy to improve the utilization of active sites of organic electrode materials in lithium-ion batteries.
O) of phthalocyanine is more exposed and the utilization of active sites for embedding lithium ions are greatly enhanced. At the same time, the large surface area is beneficial to the contact between materials and the electrolyte, which results in a faster transmission path. As a result, the first discharge specific capacities of the graphene composite phthalocyanine (TN-MPc@GN, M = Fe, Co, Ni) electrodes are 440.8, 765.0 and 753.9 mA h g−1, respectively. After 420 cycles, the specific capacities increased to 734.6, 844.7 and 949.1 mA h g−1, respectively, which are much higher than those of the pristine three phthalocyanine (TN-MPC, M = Fe, Co, Ni) electrodes without the graphene composite. Using the graphene composite can be an effective strategy to improve the utilization of active sites of organic electrode materials in lithium-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...