Mechanism of galvanic reduction of selenate oxyanions and surface immobilization by nano zero-valent iron aggregates under anaerobic conditions: towards high electron efficiency†
Abstract
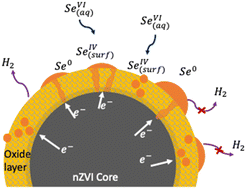
Selenate(VI) – as opposed to selenite(IV) – species encountered in industrial effluent water pose challenges to their effective elimination due to poor adsorption and chemical reduction response. Nano zero-valent iron (nZVI) is a strong reducing agent that shows a lot of promise in reducing selenate to low ppb levels. However, this is achieved with a large excess of nZVI that renders its application non-sustainable. In this work the galvanic mechanism of selenate reduction on the nZVI surface is studied with the view of increasing the electron efficiency/selectivity and thereby ultimately the efficacy of the process. Under anaerobic conditions, nZVI, consisting of chains of 60 ± 20 nm size spherical particles, forms upon immersion in water with ∼3 nm thick shells of predominantly Fe(II) hydrous oxides. Experiments were conducted to determine the kinetics and nature of Se(VI) uptake on nZVI at pH 9 and 22 °C by performing nanoscale surface characterization. It is proposed that selenate (SeO42−(aq)) converts to selenite (SeO32−(surf)) via reductive adsorption on the hydrous oxide surface of nZVI by electron exchange with Fe(II) yielding inner-sphere selenite complexes anchored on the resultant mixed Fe(II/III) surface layer. The chemically adsorbed Se(IV) is reduced galvanically to Se(0) by metallic iron core electrons conducted through the Se-enriched Fe(0/II/III) oxide layer. It is postulated that semiconducting Se0 nanodots nucleating onto and into the surface oxide layer play a critical role in enhancing the selective reduction of selenium (down to a few ppb), while suppressing the parasitic evolution of hydrogen leading to record electron efficiency as high as 70%.


 Please wait while we load your content...
Please wait while we load your content...