Comparing energy demands and longevities of membrane-based capacitive deionization architectures†
Abstract
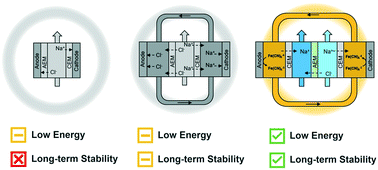
Several capacitive deionization (CDI) cell architectures employ ion-exchange membranes to control the chemistry of the electrolyte contacting the electrodes. Here, we experimentally examined how exposing carbon electrodes to either a saline electrolyte or an electrolyte containing a soluble redox-active compound influenced deionization energy demands and long-term stability over ∼50 hours. We specifically compared the energy demands (W h L−1) required to deionize 20 mM NaCl to 15 mM with a 50% water recovery as a function of productivity (L m−2 h−1). Relative to a conventional membrane capacitive deionization (MCDI) cell, flowing saline electrolyte over the electrodes did not affect energy demands but increased electrode salt adsorption capacities and capacity retention over repeated cycles. Exposing the electrodes to an electrolyte containing a redox-active compound, which made the cell behave similarly to an electrodialysis system, dramatically reduced energy demands and showed remarkable stability over 50 hours of operation. These experimental results indicate that using a recirculated soluble redox-active compound in the electrolyte contacting the electrodes to balance charge leads to far more energy efficient brackish water deionization than when charge is balanced by the electrodes undergoing capacitive charging/discharging reactions.


 Please wait while we load your content...
Please wait while we load your content...