Nitrogen-doped carbon nanotube encapsulated Fe7S8 nanoparticles for the high-efficiency and selective removal of Pb2+ by pseudocapacitive coupling†
Abstract
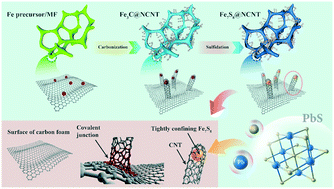
Lead (Pb) is one of the most common public hazards in drinking water, and it widely derives from mining, batteries, the waste from dye manufacturing, and so on. To eliminate this critical environmental issue, capacitive deionization (CDI) technology has been recognized as a potential alternative for the highly-efficient capture of Pb ions, owing to its operational convenience, environmental-friendliness, and low cost. In this work, we utilized a melamine foam frame to prepare nitrogen-doped carbon nanotube encapsulated Fe7S8 nanoparticles (Fe7S8@NCNT) via three hydrothermal, carbonization, and sulfidation steps for the pseudocapacitive removal of Pb2+ ions from water. The synergistic effect derived from Fe2+/Fe3+ redox pairs and specific structural design endowed the Fe7S8@NCNT electrode with excellent pseudocapacitance and enhanced CDI performance in comparison with Fe3C@NCNT and Fe7S8@NF. As a result, the exceptional Pb2+ removal capacity reached an experimental maximum value of 223.1 mg g−1 at 1.2 V with superior cycling stability. The formation of a Pb–S band after the pseudocapacitive coupling of Pb2+ ions on the surface of the electrode was confirmed based on ex situ Raman and XPS studies, suggesting a deep pseudocapacitance electrosorption mechanism for Pb2+ ions. Furthermore, Fe7S8@NCNT showed high selectivity toward Pb2+ under competition from ubiquitous Na+ and Ca2+ ions at relatively high concentrations, considering practical applications, and this is also supported by density functional theory (DFT) calculations. Thus, Fe7S8@NCNT can show great promise as a cathode for removing heavy metal Pb2+ ions from water.
- This article is part of the themed collection: Environmental Remediation


 Please wait while we load your content...
Please wait while we load your content...