π-Delocalization in phosphaphthalimide and its ambident reactivity (O/P) toward main-group electrophiles†
Abstract
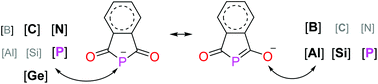
The report on phosphaphthalimide (1), the phosphorus analogue of the phthalimide anion, dates back to forty years ago. However, the presence of π-delocalization between two-coordinated phosphorus centre and neighbouring carbonyl groups in 1 has been underestimated. Herein, sodium salts of 1 were obtained through a convenient procedure on a relatively large scale with a modified procedure. Reactivity studies demonstrated that 1 is indeed a good electrophile and the essential role of π-delocalization in 1 controlling its ambident properties. NBO analysis revealed the p–π conjugation and p–σ* hyperconjugation in 1 affecting its bond lengths in opposite ways.


 Please wait while we load your content...
Please wait while we load your content...