Enhanced Ni–Ce interactions to enable efficient low-temperature catalytic CO2 methanation†
Abstract
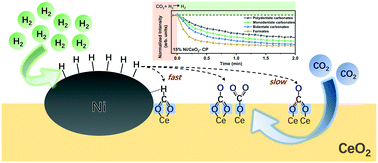
Metal–support interfacial sites have been widely employed as active sites for CO2 hydrogenation to enhance the catalytic performance. However, experimental identification of interfacial sites and determination of the origin of the activity enhancement by the interfacial sites remain a challenge. In this study, Ni/CeO2 catalysts were synthesized via a continuous co-precipitation method in a T-shaped microchannel mixer to construct abundant Ni–CeO2 interfaces. Compared to Ni/CeO2 catalysts prepared by the conventional impregnation method, catalysts synthesized by the improved co-precipitation method showed much better activity and stability for CO2 hydrogenation to methane, enabling efficient low-temperature catalytic CO2 methanation. A series of structural characterization and temperature-programmed experiments revealed that the improved co-precipitation method promoted the formation of Ni–O–Ce interfacial sites, providing more number of active sites for H2-assisted CO2 activation and thereafter leading to an improved CO2 methanation performance. This study provides an insight into interfacial sites in heterogeneous catalytic processes.


 Please wait while we load your content...
Please wait while we load your content...