Facet-dependent CO2 reduction reactions on kesterite Cu2ZnSnS4 photo-electro-integrated electrodes†
Abstract
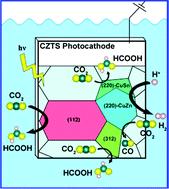
Photoelectrochemical CO2 reduction by Cu2ZnSnS4 (CZTS) photocathodes is a potentially low-cost and high-efficiency CO2 conversion approach. However, the current CZTS-based photocathodes for the CO2 reduction reaction (CO2RR) are challenged by the active side reaction of the hydrogen evolution reaction (HER) and the incompatibility with efficient electrocatalysts. In this work, by means of density functional theory (DFT), we predict that a (220)-facet-suppressed kesterite CZTS could be an efficient photo-electro-integrated photocathode for formic acid production in the CO2RR. The results show that the competitive HER is mostly favored on the (220) facet. And the CO2RR for formic acid production on the (112) and (312) facets exhibits a thermodynamic energy barrier lower than 0.26 eV. Different from the d-band theory in metal electrocatalysts, it is found that the density of low energy unoccupied states in the S 3p orbital plays a key role in determining the CO2RR reaction path of the kesterite CZTS. Furthermore, two different trends of adsorption energy depending on the chemical characteristic of adsorbates are analyzed. Our study unveils the potential for selectively reducing CO2 into formic acid with kesterite CZTS and provides a possible route for manipulating the electrocatalytic properties of metal sulfide catalysts.


 Please wait while we load your content...
Please wait while we load your content...