Influence of intermolecular interactions on the infrared complex indices of refraction for binary liquid mixtures†
Abstract
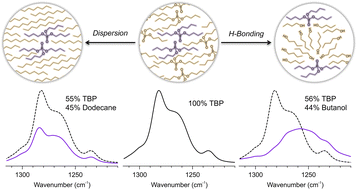
This paper investigates the accuracy of deriving the composite optical constants of binary mixtures from only the complex indices of refraction of the neat materials. These optical constants enable the reflectance spectra of the binary mixtures to be modeled for multiple scenarios (e.g., different substrates, thicknesses, volume ratios), which is important for contact and standoff chemical detection. Using volume fractions, each mixture's complex index of refraction was approximated via three different mixing rules. To explore the impact of intermolecular interactions, these predictions are tested by experimental measurements for two representative sets of binary mixtures: (1) tributyl phosphate combined with n-dodecane, a non-polar medium, to represent mixtures which primarily interact via dispersion forces and (2) tributyl phosphate and 1-butanol to represent mixtures with polar functional groups that can also interact via dipole–dipole interactions, including hydrogen bonding. The residuals and the root-mean-square error between the experimental and calculated index values are computed and demonstrate that for miscible liquids in which the average geometry of the cross-interactions can be considered isotropic (e.g., dispersion), the refractive indices of the mixtures can be modeled using composite n and k values derived from volume fractions of the neat liquids. Conversely, in spectral regions where the geometry of the cross-interactions is more restricted and anisotropic (e.g., hydrogen bonding), the calculated n and k values vary from the measured values. The impact of these interactions on the reflectance spectra are then compared by modeling a thin film of the binary mixtures on an aluminum substrate using both the measured and the mathematically computed indices of refraction.


 Please wait while we load your content...
Please wait while we load your content...