Green syntheses of stable and efficient organic dyes for organic hybrid light-emitting diodes†
Abstract
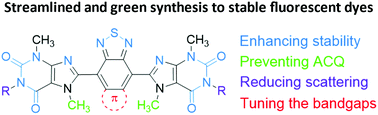
Organic hybrid light-emitting diodes (hybrid-LEDs) employ organic dyes as light converters on top of commercial blue inorganic LEDs, replacing incumbent inorganic phosphor light converters synthesized from rare-earth and/or toxic metallic elements to optimize device environmental sustainability. Here, we present two naturally derived organic dyes for hybrid-LEDs, highlighting stability and efficiency enhancement based on a novel “acceptor–acceptor” molecular design. This “acceptor–acceptor” skeleton comprises theobromine and thiadiazole, two electron-withdrawing groups that lower energy levels and suppress photooxidation. This differentiates these dyes from the widely adopted “donor–acceptor” skeleton, where photooxidation is facilitated by the presence of electron-donating units. Simultaneously, sidechains on organic dyes used to enhance solution processability, crucial for film transparency, introduce an additional photooxidation pathway. With this “acceptor–acceptor” skeleton, the destabilization from sidechains was offset by the stability enhancement from the electronic effects in the backbone. When blended within an industrial polymer, poly(styrene-butadiene-styrene) (SBS), their enhanced solubility enables the formation of highly transparent films, crucial for reducing scattering loss in LEDs. Furthermore, resultant dye-SBS films achieved photoluminescence quantum yields (PLQYs) of around 90% under ambient conditions. Taking advantage of their transparency and solution processability, we fabricated a waveguide with this theobromine-dye-SBS composite, which was subsequentially assembled into an edge-lit LED device of no glare and enhanced aesthetics.


 Please wait while we load your content...
Please wait while we load your content...