Crystalline mixed-valence copper supramolecular isomers for electroreduction of CO2 to hydrocarbons†
Abstract
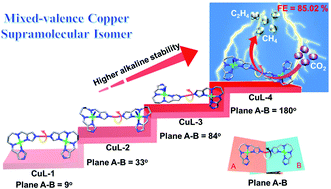
Supramolecular isomers can serve as effective model systems to explore the stable self-assembly process of specific structures, but it is often difficult to capture multiple variants in different stable states. Here, four variants (CuL-1, CuL-2, CuL-3, and CuL-4) assembled from the same CuICuIIL-metalloligand motif form different structures in which the dihedral angles of adjacent CuICuIIL-metalloligands (9°, 33°, 84°, 180°) increase gradually. CuL-2, CuL-3 and CuL-4 belong to supramolecular isomers. The relevant experimental and calculative results demonstrate that the larger the dihedral angle of adjacent CuICuIIL-metalloligands, the higher the stability of the structural variant. Moreover, the metastable structures (CuL-1, CuL-2, and CuL-3) can undergo single crystal-to-single crystal transformation into the most stable variant (CuL-4). Additionally, based on the unique mixed-valence copper structure distribution characteristics and strong alkali stability, CuL-4 is applied as an electrocatalyst for CO2 electroreduction (CO2RR), in which CO2 can be reduced to high-value hydrocarbons with a total faradaic efficiency of 85.02% (FECH4 = 67.76%, FEC2H4 = 17.26%) at −1.0 V (j = 477.42 mA cm−2). As far as we know, CuL-4 is better than most crystalline CO2RR catalysts. Furthermore, the corresponding DFT calculation proves that the independent Cu(I) sites are mainly responsible for CH4 generation, while the synergistic two adjacent Cu(II) sites are inclined to produce C2H4. Significantly, crystalline CuL-4 can serve as a well-defined model electrocatalyst to explore the role of evenly distributed mixed-valence copper active sites in reducing CO2 to hydrocarbons.


 Please wait while we load your content...
Please wait while we load your content...