The role of complementary shape in protein dimerization
Abstract
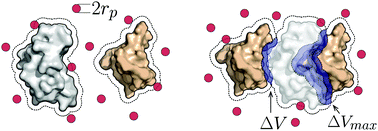
Shape guides colloidal nanoparticles to form complex assemblies, but its role in defining interfaces in biomolecular complexes is less clear. In this work, we isolate the role of shape in protein complexes by studying the reversible binding processes of 46 protein dimer pairs, and investigate when entropic effects from shape complementarity alone are sufficient to predict the native protein binding interface. We employ depletants using a generic, implicit depletion model to amplify the magnitude of the entropic forces arising from lock-and-key binding and isolate the effect of shape complementarity in protein dimerization. For 13% of the complexes studied here, protein shape is sufficient to predict native complexes as equilibrium assemblies. We elucidate the results by analyzing the importance of competing binding configurations and how it affects the assembly. A machine learning classifier, with a precision of 89.14% and a recall of 77.11%, is able to identify the cases where shape alone predicts the native protein interface.
- This article is part of the themed collection: Soft Matter Editor-in-Chief's Choice


 Please wait while we load your content...
Please wait while we load your content...