Discovery of phosphotyrosine-binding oligopeptides with supramolecular target selectivity†
Abstract
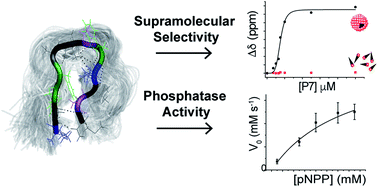
We demonstrate phage-display screening on self-assembled ligands that enables the identification of oligopeptides that selectively bind dynamic supramolecular targets over their unassembled counterparts. The concept is demonstrated through panning of a phage-display oligopeptide library against supramolecular tyrosine-phosphate ligands using 9-fluorenylmethoxycarbonyl-phenylalanine-tyrosine-phosphate (Fmoc-FpY) micellar aggregates as targets. The 14 selected peptides showed no sequence consensus but were enriched in cationic and proline residues. The lead peptide, KVYFSIPWRVPM-NH2 (P7) was found to bind to the Fmoc-FpY ligand exclusively in its self-assembled state with KD = 74 ± 3 μM. Circular dichroism, NMR and molecular dynamics simulations revealed that the peptide interacts with Fmoc-FpY through the KVYF terminus and this binding event disrupts the assembled structure. In absence of the target micellar aggregate, P7 was further found to dynamically alternate between multiple conformations, with a preferred hairpin-like conformation that was shown to contribute to supramolecular ligand binding. Three identified phages presented appreciable binding, and two showed to catalyze the hydrolysis of a model para-nitro phenol phosphate substrate, with P7 demonstrating conformation-dependent activity with a modest kcat/KM = 4 ± 0.3 × 10−4 M−1 s−1.


 Please wait while we load your content...
Please wait while we load your content...