HSA-Lys-161 covalent bound fluorescent dye for in vivo blood drug dynamic imaging and tumor mapping†
Abstract
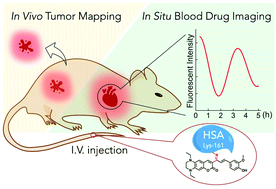
The specific combination of human serum albumin and fluorescent dye will endow superior performance to a coupled fluorescent platform for in vivo fluorescence labeling. In this study, we found that lysine-161 in human serum albumin is a covalent binding site and could spontaneously bind a ketone skeleton quinoxaline–coumarin fluorescent dye with a specific turn-on fluorescence signal for the first time. Supported by the abundant drug binding domains in human serum albumin, drugs such as ibuprofen, warfarin and clopidogrel could interact with the fluorescent dye labeled human serum albumin to feature a substantial enhancement in fluorescence intensity (6.6-fold for ibuprofen, 4.5-fold for warfarin and 5-fold for clopidogrel). The drug concentration dependent fluorescence intensity amplification realized real-time, in situ blood drug concentration monitoring in mice, utilizing ibuprofen as a model drug. The non-invasive method avoided continuous blood sample collection, which fundamentally causes suffering and consumption of experimental animals in the study of pharmacokinetics. At the same time, the coupled fluorescent probe can be efficiently enriched in tumors in mice which could map a tumor with a high-contrast red fluorescence signal and could hold great potential in clinical tumor marking and surgical resection.
- This article is part of the themed collection: Most popular 2021 analytical chemistry articles, 2021


 Please wait while we load your content...
Please wait while we load your content...