Identification of a covalent binder to the oncoprotein gankyrin using a NIR-Based OBOC screening method†
Abstract
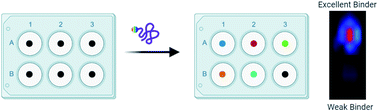
Despite huge advancements in the process of synthesizing small molecules as part of one-bead-one-compound (OBOC) libraries, progress lags in the ability to screen these libraries against proteins of interest. Recently, we developed a method to screen OBOC libraries in which a target protein is labeled with a near-infrared (NIR) range fluorophore. The labeled protein incubates with beads of a library in a 96-well plate, then the plate is imaged for fluorescence. Fluorescence intensities produced by the labeled protein binding the bead can be quantitated and provide a basis to rank hits. Here, we present an application of this technique by screening the oncoprotein gankyrin against a 343-member peptoid library. The library was composed of four positions occupied by one of seven amines. In the third position, an amine that facilitates covalent binding via a sulfonyl fluoride moiety was incorporated. After screening for gankyrin binders twice, ten structures showed overlap in the types of amines present at each position. These initial hits were validated with an in-gel fluorescence assay in which the labeled ligands covalently interacted with purified gankyrin. Excitingly, one peptoid was validated from this analysis. This hit was also shown to bind gankyrin in the presence of HEK 293T lysate. Results from this study demonstrate successful use of our screening method to quickly identify quality binders to a target protein of interest.
- This article is part of the themed collection: Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...