Lithium–bromine exchange reaction on C60: first theoretical proposal of a stable singlet fullerene carbene without the heteroatom†‡
Abstract
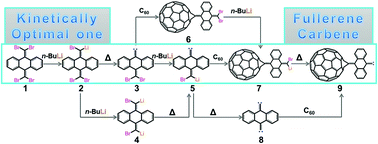
Thus far, all the stable singlet carbenes consist of a center carbon atom, and electron donor or withdrawing substituents with heteroatoms, such as, N, P, O, and S. Herein, carbene was generated by the lithium–bromine exchange reaction and used to functionalize C60. The reaction mechanisms, including the lithium–bromine exchange and carbene addition reactions, were studied in detail with ab initio density functional theory. The interaction between LiBr and the carbon atom was first characterized as a mainly ionic interaction, combined with the coordinate interaction and hyperconjugation. Then, a novel stable singlet fullerene carbene without the heteroatom, was primitively generated via lithium–bromine exchange and carbene addition reactions; it exhibited a remarkable stability with high singlet–triplet energy gap and HOMO–LUMO energy gap. This computational survey indicates that fullerene carbene will be a potential ligand in the coordinated compounds, and is useful for further functionalization and applications of fullerenes. Furthermore, theoretical results show that the lithium–bromine exchange reaction would be a powerful and feasible tool to functionalize the double bonds in some aliphatic and aromatic olefins, including anthracene, benzene, propylene, ethylene, 1-butene, propanal, and 3-pentanone. Based on the carbene reaction mechanisms and nature of corresponding bonds in their products, two indexes, bond length and the hybridization state of atoms, were suggested to distinguish the carbene addition and insertion to the double bond.


 Please wait while we load your content...
Please wait while we load your content...