Antioxidant product analysis of Hulu Tea (Tadehagi triquetrum)†
Abstract
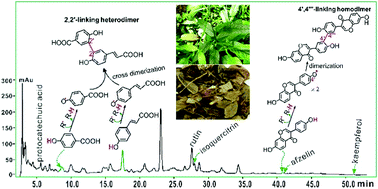
The dried plant of Tadehagi triquetrum is called Hulu Tea in traditional Chinese medicine (TCM). This study attempted to analyze the antioxidant products of the interaction with cellular reactive oxygen species. LAEH, a lyophilized aqueous extract of Hulu Tea, was prepared and analyzed using high-performance liquid chromatography. Eight phytophenols were found, namely, rutin, isoquercitrin, p-coumaric acid, p-hydroxybenzoic acid (pHBA), kaempferol, protocatechuic acid, afzelin, and daidzein. The antioxidant levels of the phytophenols were evaluated using the 2-phenyl-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide (PTIO˙) trapping, 1,1-diphenyl-2-picryl-hydrazl (DPPH˙) trapping, Fe3+-reducing, and Cu2+-reducing assays. The reaction products of DPPH˙ with phytophenols were further analyzed using ultra-performance liquid chromatography coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry (UPLC-ESI-Q-TOF-MS/MS). A series of molecular ion and fragment peaks were observed, which corresponded to seven phytophenol–DPPH adducts, four phytophenol–phytophenol homodimers, and one p-coumaric acid–pHBA heterodimer. Seven possible heterodimer linking sites were further calculated using computational chemistry based on density functional theory with the methanol solvation model at the B3LYP (or UB3LYP)-D3/6-311+G(d,p) level. However, the 2,2′-linking heterodimer possessed the lowest enthalpy change (ΔH) value of −69.54 kcal mol−1; the 2,2′-bond presented a Laplacian bond order value of 1.083 and a bond length of 148.9 pm. In conclusion, the eight antioxidant phytophenols can produce three types of antioxidant products, phytophenol–radical adduct, phytophenol–phytophenol homodimer, and phytophenol–phytophenol heterodimer. The phytophenol–phytophenol heterodimer refers to the p-coumaric acid–pHBA 2,2′-linking heterodimer rather than the 2-8′ benzofuran p-coumaric acid/pHBA heterodimer.


 Please wait while we load your content...
Please wait while we load your content...