Post-synthesis modification of functionalised polyhedral oligomeric silsesquioxanes with encapsulated fluoride – enhancing reactivity of T8-F POSS for materials synthesis†
Abstract
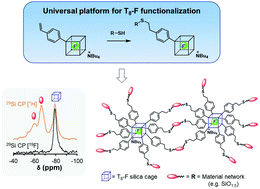
Previous studies have demonstrated that functionalised T8 polyhedral oligomeric silsesquioxanes with fluoride anions encapsulated within the T8 cage (T8-F) have very low reactivity, with no reports of successful modification of the organic moieties. Herein, we report the successful modification of a styryl functionalised T8-F compound with mercaptopropyltriethoxysilane using a thiol–ene click reaction. This strategy enables the vinyl moiety to be modified while maintaining the influence of the electron-withdrawing phenylene group, which stabilises the fluoride within the cage. The triethoxysilylated T8-F cages were subsequently converted into nanohybrids by sol–gel processing. Multinuclear NMR experiments (29Si, 19F, 13C) demonstrated that the POSS cage remained intact during both post functionalisation of T8-F and subsequent sol-gel processing. Furthermore, 29Si cross polarisation solid state NMR experiments using 19F as a cross polarisation agent demonstrated that the fluoride anions remain localised within the cages in the nanohybrids. The functionalisation of the styryl bearing T8-F POSS cage is the first example of both the successful modification of the poorly reactive T8-F family and the formation of a nanohybrid material incorporating fluoride ions immobilised within covalently bound T8 cages.


 Please wait while we load your content...
Please wait while we load your content...