Time-resolved SANS reveals pore-forming peptides cause rapid lipid reorganization†
Abstract
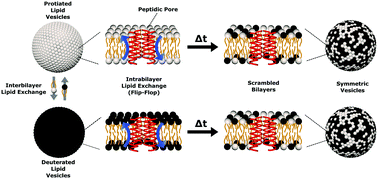
Cells depend on proper lipid transport and their precise distribution for vital cellular function. Disruption of such lipid organization can be initiated by external agents to cause cell death. Here, we investigate two antimicrobial pore-forming peptides, alamethicin and melittin, and their influence on lipid intervesicular exchange and transverse lipid diffusion (i.e. flip-flop) in model lipid vesicles. Small angle neutron scattering (SANS) and a strategic contrast matching scheme show the mixing of two isotopically distinct dimyristoylphosphocholine (DMPC) vesicle populations is promoted upon the addition of high (1/40) and low (1/150, 1/1000) peptide-to-lipid (P/L) molar ratios. Parsing out the individual exchange and flip-flop rate constants revealed that alamethicin increases both DMPC flip-flop and exchange by ≈2-fold when compared to methanol alone (the carrier solvent of the peptides). On the other hand, melittin affected DMPC flip-flop by a factor of 1 to 4 depending on the concentration, but had little effect on inter-vesicle lipid exchange at low P/L ratios. Thermodynamic parameters measured at high protein concentrations (P/L = 1/40) yielded remarkable similarity in the values obtained for both peptides, indicating likeness in their mechanism of action on lipid motion despite differences in their proposed oligomeric pore structures. The entropic contributions to the free energy of activation became favorable upon peptide addition, while the enthalpy of activation remained the major barrier to lipid exchange and flip-flop.
- This article is part of the themed collection: NJC Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...