Key intermediates from simultaneous removal of NOx and chlorobenzene over a V2O5–WO3/TiO2 catalyst: a combined experimental and DFT study†
Abstract
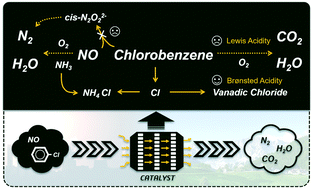
Simultaneous elimination of NOx and dioxins in a selective catalytic reduction (SCR) converter has been proved to be a promising technology for multipollutant control in stationary emission. Lack of a systematic understanding of the complex interaction mechanism between a SCR reaction and catalytic oxidation is one of the main challenges of this technology. Herein, using chlorobenzene (CB) as the model molecule of dioxins, we perform an in-depth study on the interaction mechanism and key intermediates over a V2O5–WO3/TiO2 catalyst via in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) and density functional theory (DFT) calculations. The results indicate that CB suppresses the active cis-N2O22− formation due to the enhancement of the reducibility of V oxides. The dissociated Cl− combines with vanadium to form vanadium chloride which activates adjacent bridging hydroxyl groups, resulting in the enhancement of surface Brønsted acidity. Besides, the presence of NO and O2 is conducive to CB deep oxidation while NH3 exhibits an inhibitory effect.


 Please wait while we load your content...
Please wait while we load your content...