The interplay of Ag and ferromagnetic MgFe2O4 for optimized oxygen-promoted hydrogen evolution via formaldehyde reforming†
Abstract
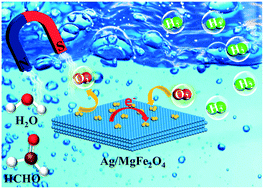
Hydrogen is one of the most promising alternative energy resources due to its environmental benignity and high energy density. However, the development of high performance catalysts for efficient and stable liquid-phase hydrogen generation remains a major challenge for the widespread use of hydrogen energy. Here, Ag supported on ferromagnetic MgFe2O4 nanoparticles (Ag/MgFe2O4) was employed as an efficient, stable, and magnetoswitchable catalyst for the conversion of formaldehyde solution into H2 using O2 as a cocatalyst at room temperature. It was found that the adsorption and activation of O2 on the Ag surface can be enhanced by the abundant surface oxygen vacancies on MgFe2O4 support and the electron transfer between MgFe2O4 and Ag, leading to the generation of the main active species ˙OOH radicals and the subsequent cleavage of C–H bonds in HCHO to realize hydrogen evolution. Meanwhile, magnetic control experiments are designed to switch the catalytic reactions between “on” and “off” states, resulting in the effective recovery and reuse of the catalysts. The present research introduces a new means to create a magnetoswitchable catalytic system for scalable hydrogen energy applications.


 Please wait while we load your content...
Please wait while we load your content...