Vacuum ultraviolet photochemistry of sulfuric acid vapor: a combined experimental and theoretical study†
Abstract
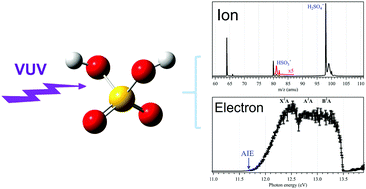
We present a vacuum ultraviolet (VUV) photoionization study of the gas-phase sulfuric acid (H2SO4) molecule in the 11–14 eV energy range by using the method of synchrotron radiation-based double imaging photoelectron photoion coincidence (i2PEPICO) spectroscopy complemented with accurate theoretical calculations. The slow photoelectron spectrum (SPES) of H2SO4 has been acquired and the three electronic states of H2SO4+, X2A, A2A and B2A have been populated and assigned. The adiabatic ionization energy of the H2SO4 molecule towards the X2A cationic ground state is measured at 11.684 ± 0.006 eV, in accordance with high-level calculated findings. With increasing photon energies, the H2SO4+ cation dissociates into HSO3+ and OH fragments and their adiabatic appearance energy is measured at 13.498 ± 0.007 eV. Then, the enthalpies of formation for the species involved in the photoionization and dissociative photoionization have been determined through a thermochemical cycle.
- This article is part of the themed collection: Festschrift Ivan Powis: Advances in Molecular Photoelectron Spectroscopy: Fundamentals & Application


 Please wait while we load your content...
Please wait while we load your content...