Electrolyte buffering species as oxygen donor shuttles in CO electrooxidation†
Abstract
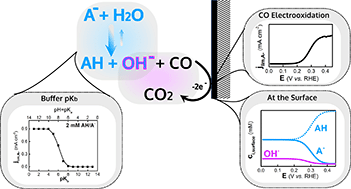
Electrolyte buffering species have been shown to act as proton donors in the hydrogen evolution reaction (HER). Analogously, we study here whether these electrolyte species may participate in other reactions by investigating CO electrooxidation (COOR) on a gold rotating disk electrode. This model system, characterized by fast kinetics, exhibits a diffusion-limited regime, which helps in the identification of the species dictating the diffusion-limited current. Through a systematic concentration dependence study in a variety of buffers, we show that electrolyte buffering species act as oxygen donor shuttles in COOR, lowering the reaction overpotential. A similar correlation between electrolyte and electrocatalytic activity was observed for COOR on a different electrode material (Pt). Probing the electrode–electrolyte interface by attenuated total reflection infrared spectroscopy (ATR-FTIR) and modelling the surface speciation to include the effect of the solution reactions, we propose that the buffer conjugated base generates the oxygen donor (i.e. OH−) through its acid–base reaction with water.
- This article is part of the themed collection: 2021 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...