Born to be different: the formation process of Cu nanoparticles tunes the size trend of the activity for CO2 to CH4 conversion†
Abstract
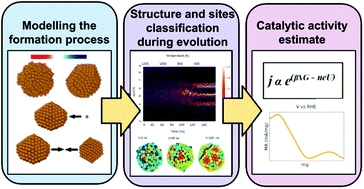
We investigate the impact of the formation process of Cu nanoparticles on the distribution of adsorption sites and hence on their activity. Using molecular dynamics, we model formation pathways characteristic of physical synthesis routes as the annealing of a liquid droplet, the growth proceeding via the addition of single atoms, and the coalescence of individual nanoparticles. Each formation process leads to different and characteristic size-dependent distributions of their adsorption sites, catalogued and monitored on-the-fly by means of a suitable geometrical descriptor. Annealed or coalesced nanoparticles present a rather homogeneous distribution in the kind and relative abundance of non-equivalent adsorption sites. Atom-by-atom grown nanoparticles, instead, exhibit a more marked occurrence of adsorption sites corresponding to adatoms and small islands on (111) and (100) facets. Regardless of the formation process, highly coordinated sites are more likely in larger nanoparticles, while the abundance of low-coordination sites depends on the formation process and on the nanoparticle size. Furthermore, we show how each characteristic distribution of adsorption sites reflects in different size trends for the Cu-nanoparticle activity, taking as an example the electro-reduction of CO2 into CH4. To this end, we employ a multi-scale method and observe that the faceted but highly defected structures obtained during the atom-by-atom growth become more and more active with increasing size, with a mild dependence on the original seed. In contrast, the activity of Cu-nanoparticles obtained by annealing decreases with their size, while coalesced nanoparticles’ activity shows a non-monotonic behaviour.


 Please wait while we load your content...
Please wait while we load your content...