Photoactive layers for photovoltaics based on near-infrared absorbing aryl-substituted naphthalocyanine complexes: preparation and investigation of properties†
Abstract
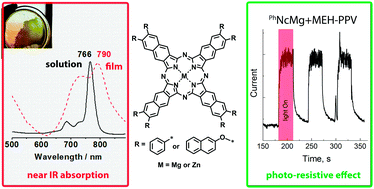
Photoactive layers based on aryl- and aryloxy-substituted naphthalocyanines and conductive polymer poly[2-methoxy-5-(2′-ethylhexyloxy)-1,4-phenylene vinylene] (MEH-PPV) were prepared using the spin-coating technique and their conductivity was tested in dark and under illumination. For this purpose novel octa-2-naphthoxy-substituted naphthalocyanines were synthesized starting from 6,7-di(2-naphthoxy)naphthalene-2,3-dicarbonitrile. For those novel naphthalocyanine complexes, spectral and electrochemical data were measured and compared with corresponding ones for other aryl-substituted analogues. In comparison to the previously studied naphthalocyanines with alkyl- and phenyl-groups, the formal oxidation and reduction potentials were rather similar. All target complexes demonstrate intense near-infrared absorption at 760–790 nm, which is about 30 nm bathochromically shifted in thin films. The photo-resistive effect was found to increase from composites comprised of naphthoxy- to phenyl-substituted naphthalocyanines. This peculiarity was explained by using optical and atomic force microscopy in terms of different sizes of the aggregates formed. The photo-response time for the novel composites was approximately 3 s, which is about 20 times faster than measured previously for the films deposited via the drop-casting technique.


 Please wait while we load your content...
Please wait while we load your content...