Adsorption of bovine serum albumin on gold nanoprisms: interaction and effect of NIR irradiation on protein corona†
Abstract
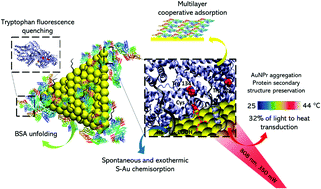
Because of their photothermal properties, gold nanoparticles (AuNPs) have gained attention regarding their use in drug delivery and therapeutic applications. In this sense, it is interesting to consider their interactions with biologically available proteins, such as serum albumin, as well as the effects of irradiation and photothermal conversion on the protein structure that can lead to a loss of function or generate an immune response. Gold nanoprisms (AuNPrs) have gained interest due to their low toxicity, ease of synthesis, and excellent stability, promoting their use in bioapplications such as surface-enhanced Raman spectroscopy (SERS), drug delivery, and photothermal therapy. The interaction between AuNPrs, with plasmon bands centred in the near-infrared region (NIR), and bovine serum albumin (BSA) has not been explored yet. UV-Vis spectroscopy, dynamic light scattering (DLS) and fluorescence spectroscopy were used to study the interaction between AuNPrs and BSA in addition to estimation of the adsorption rate and kinetic and thermodynamic parameters (K, ΔH°, ΔG°, ΔS°, and Ea) using adsorption isotherms and Langmuir and Freundlich models. The results suggest spontaneous cooperative binding in multilayer adsorption, achieved by the chemisorption of BSA on the AuNPr surface through the S–Au interaction, as confirmed by Raman spectroscopy. On the other hand, the photothermal conversion efficiency (PE) of the coated nanoparticles after NIR irradiation was assessed, resulting in a slight decrease in the PE of BSA coated on AuNPrs in comparison with that of noncapped nanoparticles. The effect of the irradiation on the protein conformation of capped nanoparticles was also assessed; circular dichroism showed BSA unfolding upon interaction with AuNPrs, with a decrease in the α-helix and β-sheet contents, as well as an increase in random coil conformations. Changes in the Raman spectrum suggest a modification of the disposition of the protein residues exposed to the gold surface after NIR irradiation; but at the secondary structure level, no relevant changes were observed. This provides possibilities for the use of NPs–BSA for bioapplications based on the photothermal effect promoted by laser irradiation, since the biological identity of the protein is preserved after NIR irradiation.
- This article is part of the themed collection: Celebrating Latin American Talent in Chemistry


 Please wait while we load your content...
Please wait while we load your content...