Influence of Ti3+ defect-type on heterogeneous photocatalytic H2 evolution activity of TiO2†
Abstract
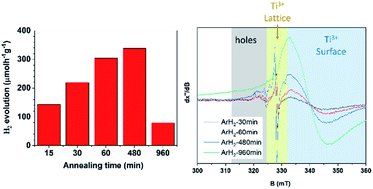
Reduced titanium dioxide has recently attracted large attention, particularly for its unique co-catalyst-free heterogeneous photocatalytic application for H2 generation. The enhanced photocatalytic activity of the reduced TiO2 was previously ascribed to the introduction of point crystal defects (mainly Ti3+ centers), which result in the formation of intrinsic co-catalytic centers and enhanced visible light absorption. In this work, we systematically investigate the effect of different defects in the TiO2−x lattice on photocatalytic H2 evolution. To introduce different types of defects, thermal annealing in air (oxidative), Ar (inert), Ar/H2 (reducing), and H2 (reducing) atmospheres was performed on commercially available anatase nanopowder. Then, the powders were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD) and high-resolution transmission electron microscopy (HR-TEM) to clarify the effect of treatment on material properties. Furthermore, the defect types were characterized by electron paramagnetic resonance (EPR) spectroscopy. We show that thermal annealing in different atmospheres can form different amounts of different defect types in the TiO2 structure. The highest photocatalytic activation is achieved by annealing the anatase powder in a reducing atmosphere for an appropriate temperature/annealing time. By combining the results from H2 generation and EPR analysis we show that the simultaneous presence of two types of defects, i.e. surface exposed Ti3+ and lattice embedded Ti3+ centers in an optimum low concentration is the determining factor for an optimized photocatalytic H2 evolution rate. In fact, annealing anatase powder under the so-reported optimized conditions in reducing atmosphere leads to the generation of a considerable amount of H2, with rates as high as 338 μmol h−1 g−1.


 Please wait while we load your content...
Please wait while we load your content...