Anesthetics significantly increase the amount of intramembrane water in lipid membranes†
Abstract
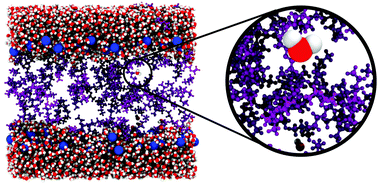
The potency of anesthesia was directly linked to the partitioning of the drug molecules in cell membranes by Meyer and Overton. Many molecules interact with lipid bilayers and lead to structural and functional changes. It remains an open question which change in membrane properties is responsible for a potential anesthetic effect or if anesthetics act by binding to direct targets. We studied the effect of ethanol, diethyl ether and isoflurane on the water distribution in lipid bilayers by combining all-atom molecular dynamics simulations and neutron diffraction experiments. The simulations show strong membrane–drug interactions with partitioning coefficients of 38%, 92% and 100% for ethanol, diethyl ether and isoflurane, respectively, and provide evidence for an increased water partitioning in the membrane core. The amount of intramembrane water molecules was experimentally determined by selectively deuterium labeling lipids, anesthetic drug and water molecules in neutron diffraction experiments. Four additional water molecules per lipid were observed in the presence of ethanol. Diethyl ether and isoflurane were found to significantly increase the amount of intramembrane water by 25% (8 water molecules). This increase in intramembrane water may contribute to the non-specific interactions between anesthetics and lipid membranes.


 Please wait while we load your content...
Please wait while we load your content...