Molecular insights into photostability of fluorinated organic photovoltaic blends: role of fullerene electron affinity and donor–acceptor miscibility†
Abstract
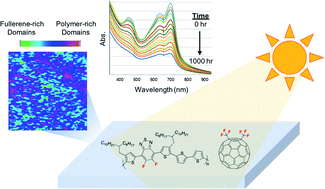
In this work, the photostability of certain organic photovoltaic (OPV) active layers was demonstrated to improve by as much as a factor of five under white light illumination in air with the use of 1,7-bis-trifluoromethylfullerene (C60(CF3)2) as the acceptor in place of PC60BM. However, the results were highly dependent on the structure and functionality within the donor material. Twelve combinations of active layer blends were studied, comprised of six different high-performance donor polymers (two fluorinated and four non-fluorinated donors) and two fullerene acceptors (PC60BM and C60(CF3)2). The relative rates of irreversible photobleaching of the active layer blends were found to correlate well with the electron affinity of the fullerene when the polymer and fullerene were well blended, but a full rationalization of the photobleaching data requires consideration of both the electron affinity of the fullerene as well as the relative miscibility of the polymer–fullerene components in the blend. Miscibility of those components was probed using a combination of time-resolved photoluminescence (TRPL) measurements and scanning tunneling microscopy (STM) imaging. The presence of fluorinated aromatic units in the donor materials tend to promote more intimate mixing with C60(CF3)2 as compared to PC60BM. The full results of these photobleaching studies and measurements of donor–acceptor miscibility, considered alongside additional photoconductance measurements and preliminary device work, provide new molecular optimization insights for improving the long-term stability of OPV active layers.


 Please wait while we load your content...
Please wait while we load your content...