Halogen-directed chemical sialylation: pseudo-stereodivergent access to marine ganglioside epitopes†‡
Abstract
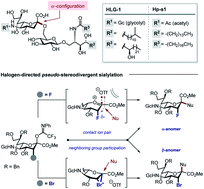
Sialic acids are conspicuous structural components of the complex gangliosides that regulate cellular processes. Their importance in molecular recognition manifests itself in drug design (e.g. Tamiflu®) and continues to stimulate the development of effective chemical sialylation strategies to complement chemoenzymatic technologies. Stereodivergent approaches that enable the α- or β-anomer to be generated at will are particularly powerful to attenuate hydrogen bond networks and interrogate function. Herein, we demonstrate that site-selective halogenation (F and Br) at C3 of the N-glycolyl units common to marine Neu2,6Glu epitopes enables pseudo-stereodivergent sialylation. α-Selective sialylation results from fluorination, whereas traceless bromine-guided sialylation generates the β-adduct. This concept is validated in the synthesis of HLG-1 and Hp-s1 analogues.
- This article is part of the themed collection: Celebrating 10 years of Chemical Science


 Please wait while we load your content...
Please wait while we load your content...