Identification, synthesis and biological evaluation of pyrazine ring compounds from Talaromyces minioluteus (Penicillium minioluteum)†
Abstract
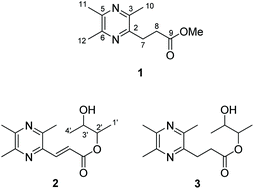
Three new pyrazine derivatives, named talaropyrazines A–C (1–3), were isolated from the chemical investigation of the fungus Talaromyces minioluteus. Their planar structures were established by extensive 1D and 2D NMR and HRESIMS spectroscopic data analyses. The absolute configurations of 2 and 3 were defined by the synthesis of all the four isomers, respectively. The detailed comparison of the NMR and HPLC spectra of the natural product and the synthetic ones allows us to propose a general mechanism for the formation of diastereomers of 2 and 3. Moreover, the in vitro bioassay showed the synthetic congener (2′R,3′R)-3 exhibited potential inhibitory activity in the murine splenocytes stimulated by anti-CD3/anti-CD28 mAbs, with IC50 values of 5.85 μM.


 Please wait while we load your content...
Please wait while we load your content...