Accessing alkyl boronic esters via visible light-mediated decarboxylative addition reactions of redox-active esters†
Abstract
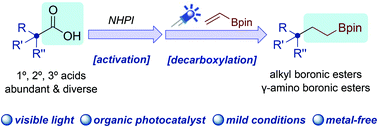
A general and facile approach for the synthesis of alkyl boronic esters via visible light-mediated decarboxylative radical addition of redox-active esters to vinyl boronic ester is presented. The reaction features mild, metal-free reaction conditions and broad substrate scope covering primary, secondary, and tertiary acids, as well as amino acids and drugs.


 Please wait while we load your content...
Please wait while we load your content...