Dual-doping of ruthenium and nickel into Co3O4 for improving the oxygen evolution activity†
Abstract
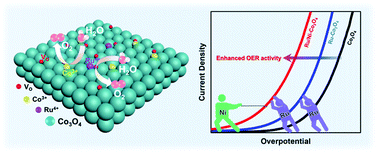
Spinel oxides Co3O4 with mixed valence cations Co2+ and Co3+ make it possible to improve the catalytic activity by regulating the distribution of cations via additional doping. Controllable synthesis of nanosized Co3O4 with excellent catalytic properties and stability towards the oxygen evolution reaction (OER) is critical for conversion and storage of sustainable source-derived electricity. Herein, Ru and Ni co-doped Co3O4 nanoparticles (Ru/Ni-Co3O4) with abundant active sites and oxygen vacancies were synthesized by a one-step hydrothermal method. Ru atoms doped into the octahedral sites improve the intrinsic activity of spinel oxide Co3O4, while the introduction of Ni creates abundant oxygen vacancies, thus exposing more active sites at the surface and accelerating the OER kinetics. The optimized Ru/Ni-Co3O4 nanoparticles possess a low overpotential (290 mV at 10 mA cm−2) and excellent stability (only 1% activity decay after 10 h), significantly superior to the state-of-the-art IrO2 and most OER electrocatalysts reported before. This work provides a new strategy to increase the density of the active sites and generate surface oxygen vacancies simultaneously without destroying the crystallinity/microstructure and use of any additional complicated treatments (such as plasma techniques and multi-step pyrolysis).
- This article is part of the themed collection: 2020 Materials Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...