Synthesis of macrocyclic poly(α-hydroxyl acids) via DABCO-mediated ROP of O-carboxylanhydrides derived from l-phenylalanine even in the presence of an alcohol†
Abstract
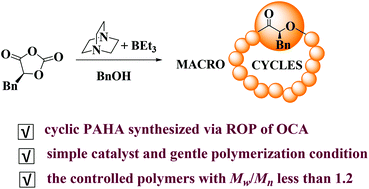
Cyclic polyesters with very interesting topologies and unique properties have great potential. However, the synthesis of cyclic polyesters via ring-opening polymerization (ROP) of O-carboxyanhydrides (OCAs) has been hardly reported to date. Herein, a simple combination of triethylboron (TEB) and 1,4-diazabicyclo-[2.2.2]octane (DABCO) in the presence of benzyl alcohol (BnOH) was explored to promote the ROP of L-phenyl O-carboxyanhydride (L-PheOCA) for the synthesis of well-defined cyclic poly(α-hydroxy acid)s (PAHAs). Such a ternary catalytic system was found to exhibit well-controlled and living catalytic activities for L-PheOCA, and the polymerization produced cyclic PAHAs with a narrow molecular weight distribution (Mw/Mn < 1.2) and the isotacticity of Pm = 0.83. Careful analysis of the catalytic system showed the interaction of DABCO, TEB, and BnOH. Combined with the kinetic experiments, it was deduced that DABCO with preferable nucleophilicity played a predominant role in the polymerization process, and BnOH acted as a cocatalyst, not as an initiator during the polymerization. The zwitterionic active species with DABCO at one end and an alkyloxide at the other end of the propagation chain was involved in the synthesis of cyclic PAHAs. The thermal properties of the resulting polymers were analyzed by DSC and TGA, respectively. The degradation temperature of cyclic PAHAs was 30 °C higher than that of the corresponding linear polymers, exhibiting much better thermal stability. This is the first work on cyclic PAHAs fabricated via ROP of OCAs, which provides one new way to synthesize cyclic PAHAs and has significant potential to enrich the diversity of cyclic PAHAs.


 Please wait while we load your content...
Please wait while we load your content...