Realising the electrochemical stability of graphene: scalable synthesis of an ultra-durable platinum catalyst for the oxygen reduction reaction†
Abstract
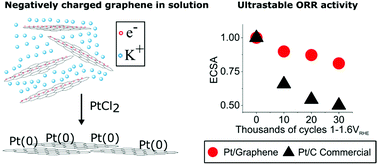
Creating effective and stable catalyst nanoparticle-coated electrodes that can withstand extensive cycling is a current roadblock in realising the potential of polymer electrolyte membrane fuel cells. Graphene has been proposed as an ideal electrode support material due to its corrosion resistance, high surface area and high conductivity. However, to date, graphene-based electrodes suffer from high defect concentrations and non-uniform nanoparticle coverage that negatively affects performance; moreover, production methods are difficult to scale. Herein we describe a scalable synthesis for Pt nanoparticle-coated graphene whereby PtCl2 is reduced directly by negatively charged single layer graphene sheets in solution. The resultant nanoparticles are of optimal dimensions and can be uniformly dispersed, yielding high catalytic activity, remarkable stability, and showing a much smaller decrease in electrochemical surface area compared with an optimised commercial catalyst over 30 000 cycles. The stability is rationalised by identical location TEM which shows minimal nanoparticle agglomeration and no nanoparticle detachment.


 Please wait while we load your content...
Please wait while we load your content...