3D ternary NixCo2−xP/C nanoflower/nanourchin arrays grown on HCNs: a highly efficient bi-functional electrocatalyst for boosting hydrogen production via the urea electro-oxidation reaction†
Abstract
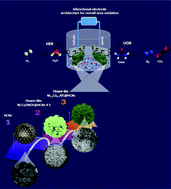
Over the last few years, substantial efforts have been made to develop earth-abundant bi-functional catalysts for urea oxidation and energy-saving electrolytic hydrogen production due to their low cost and the potential to replace traditional noble-metal-based catalysts. Nevertheless, finding a straightforward and effective route to prepare efficient catalysts with unique structural features and optimal supports still is a big challenge. Among the various candidates, metal–organic framework (MOF)-derived materials show great advantages as new kinds of active non-precious catalysts. On the other hand, the controllable integration of MOFs and carbon-based nanomaterials leads to further enhancement in terms of the stability and electrical conductivity of catalysts. In this communication, we develop an MOF-carbon-based composite to synthesize a transition metal phosphide (TMP) catalyst for the electrocatalytic oxidation of urea. First, poly(pyrrole-co-aniline) (PPCA) hollow nanospheres were fabricated via the in situ emulsion polymerization of a mixture of aniline and pyrrole in the presence of Triton X-100. Then, the simple carbonization treatment of these PPCA hollow spheres led to the carbonized hollow carbon nanospheres (HCNs) with ultrahigh surface areas and uniform nano-morphologies. After that, bimetallic MM′/MOFs (M/M′ = Ni, Co) were uniformly grown around the HCNs via a simple hydrothermal reaction (NiCo/MOF@HCNs). During the synthesis process, by adjusting Ni/Co ratios, the MOF morphology can be engineered so that by reducing the Ni/Co ratio, the flower-like structures change into urchin-like structures. Finally, this NiCo/MOF@HCNs precursor with different Ni/Co ratios during the in situ carbonization/phosphorization was chemically converted into Ni–Co mixed-metal phosphides (NixCo2−xP/C@HCNs). Finally, the electrocatalytic activity of the prepared catalysts was tested for the urea oxidation reaction (UOR) and hydrogen evolution reaction (HER).


 Please wait while we load your content...
Please wait while we load your content...