Highly active Fe–N-doped porous hollow carbon nanospheres as oxygen reduction electrocatalysts in both acidic and alkaline media†
Abstract
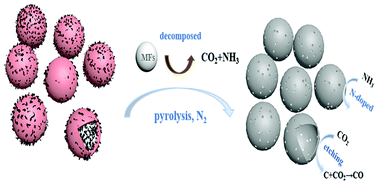
Hierarchical iron–nitrogen-codoped porous hollow carbon spheres have been synthesized by using melamine–formaldehyde (MF) resin spheres as templates, nitrogen sources and pore-forming agents. FeCl3, 1,10-phenanthroline and carbon black were used as iron, nitrogen and carbon sources. The as-obtained porous hollow carbon spheres possess a high specific surface area of 807 m2 g−1, as well as exhibited excellent electrocatalytic activity for the oxygen reduction reaction (ORR) in both acidic and alkaline media. In 0.1 M HClO4 solution, the onset potential was 0.857 V (vs. RHE) and the half-wave potential was 0.715 V, which are only 78 and 80 mV less than those of the 20% Pt/C catalyst, respectively. In addition, in 0.1 M KOH solution, the onset potential was 1.017 V and the half-wave potential was 0.871 V for the ORR, which are 22 and 28 mV more positive than those of the Pt/C catalyst, respectively. Meanwhile, the catalyst also exhibited excellent methanol tolerance and long-term durability with a more effective four-electron pathway compared to the 20% Pt/C catalyst in both acidic and alkaline media. When used as an air-cathode catalyst for a Zn–air battery, the maximum power density of a Zn–air battery with the MF-C-Fe-Phen-800 cathode was 235 mW cm−2 at a high current density of 371 mA cm−2, and a high open-circuit potential of 1.654 V, superior to that of Pt/C (199 mW cm−2, 300 mA cm−2, 1.457 V). A series of designed experiments suggested that the remarkable performance was attributed to the high specific area, hollow carbon spheres, unique hierarchical micro-mesoporous structures, high contents of pyridinic-N and graphitic-N. The superiority of the as-prepared catalyst makes it promising for use in practical applications.
- This article is part of the themed collection: 2020 Nanoscale HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...