Modulating the H-bond strength by varying the temperature for the high pressure synthesis of nitrogen rich carbon nanothreads
Abstract
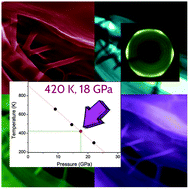
Carbon nanothreads are among the most attractive new materials produced under high pressure conditions. Their synthesis can be achieved by compressing the crystals of aromatic molecules exploiting both the anisotropic stress produced by the unidirectional applied force and that intrinsic to the crystal arrangement. We explored here the transformation of pyridine into a nitrogen rich carbon nanothread crystal by varying the pressure and temperature conditions with the twofold purpose of disclosing the microscopic mechanism of transformation and optimizing the yield and quality of the produced crystalline nanothreads. The best conditions for the synthesis were identified in the 14–18 GPa range at temperatures between 400 and 500 K with a product yield greater than 30%. The comparison of experiments performed under different P–T conditions allowed us to understand the role of high temperature, which is necessary to weaken or even destroy the complex H-bond network characterizing the pyridine crystal and preventing the correct approach of the aromatic rings for nanothread formation. X-ray diffraction data confirm the excellent 2D hexagonal packing of the nanothreads over several tens of microns, whereas the sharp absorption lines observed in the IR spectrum strongly support a substantial order along the threads. Diffraction results suggest a polytwistane structure of the threads derived from a Diels–Alder [4 + 2] polymerization involving molecules arranged in a slipped parallel configuration along the pyridine crystal a and b axes. Electron microscopy evidences an arrangement of the nanothreads in bundles of tens of nanometers.


 Please wait while we load your content...
Please wait while we load your content...