Ultrasound-assisted synthesis of BiVO4/C-dots/g-C3N4Z-scheme heterojunction photocatalysts for degradation of minocycline hydrochloride and Rhodamine B: optimization and mechanism investigation†
Abstract
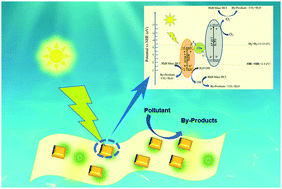
As a solid-state electron medium of composite photocatalysts, carbon dots (C-dots) increase the transition process of photogenerated carriers and inhibit their rapid recombination. In this work, novel and efficient Z-scheme heterojunction BiVO4/C-dots/g-C3N4 photocatalysts with C-dots as the photoelectron transfer center were designed. FTIR, HRTEM, TEM, XRD and PL were used to investigate the physical and chemical properties of the photocatalysts. Due to the up-conversion photoluminescence characteristics of the carbon dots, the BiVO4/C-dots/g-C3N4 photocatalysts exhibit significant interfacial charge transfer capability and a broader visible absorption range. In addition, the photocatalytic activity of the photocatalysts was improved by changing the doping amount and optimizing the synthesis conditions. As a result, the BiVO4/C-dots/g-C3N4 composites showed remarkable degradation efficiency of minocycline hydrochloride (Mino-HCl) and Rhodamine B (RhB) under near-visible LED light. Furthermore, the photodegradation efficiency of the BiVO4/C-dots/g-C3N4-40 photocatalyst was conspicuously improved over those of pure BiVO4, g-C3N4 and the composites with other content ratios. The comparative experiment also proved that the introduction of carbon dots can enhance the performance of the photocatalyst. Finally, the possible catalytic mechanism was explored via a free radical capture experiment for the active substances, and the excellent photostability and reusability of the BiVO4/C-dots/g-C3N4 composites were proved through three cycles of experiments.


 Please wait while we load your content...
Please wait while we load your content...