Separation of sodium chromate from high alkaline media based on precipitation transformation using barium hydroxide
Abstract
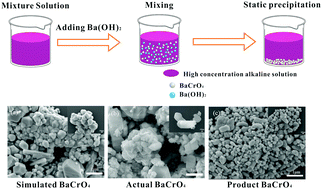
In this study, a novel precipitation transformation system, using barium hydroxide as the separation medium, was adopted to achieve the separation of sodium chromate from a concentrated NaOH solution. The alkaline solution was obtained in the liquid-phase oxidation leaching process of chromite using the so-called “sub-molten salt”. In this separation process, Na2CrO4 was transferred from liquid-phase BaCrO4 to solid-phase BaCrO4 by Ba(OH)2 precipitation. Subsequently, the effects of parameters such as reaction time, reaction temperature, material ratio, NaOH concentration, and dilution multiple on the separation behavior of sodium chromate were discussed. Then, the macroscopic kinetics of the chromium reaction in the separation system was investigated in depth. The results indicated that the new separation process is able to obviously reduce the reaction requirements, and that it has a higher separation efficiency. The conversion efficiency of sodium chromate exceeded 92% under the condition of Na2CrO4-to-Ba(OH)2 molar ratio of 1 : 1, NaOH concentration of 50 wt%, reaction time of 120 min, no dilution, and reaction temperature of 60 °C. From the perspective of the macroscopic kinetics, the apparent activation energy of the reaction process Ea = 6.870 kJ·mol−1, the apparent rate constant k = A·e−6870/RT, and the pre-exponential factor A = 0.056 min−1 were obtained. In addition, the enthalpy of activation and the entropy of activation were calculated using the Eyring equation, with ΔH≠ = 4.186 ± 2.144 kJ mol−1 and ΔS≠ = −277.863 ± 6.631 J mol−1 K−1. This study provides data support and theoretical guidance for the separation of chromate from a high-concentration alkaline sub-molten salt solution.


 Please wait while we load your content...
Please wait while we load your content...