Influence of lateral methyl/chloro substituents on the liquid crystalline and photoswitching behaviour of bent-core mesogens bearing azobenzene wings: synthesis and characterization†
Abstract
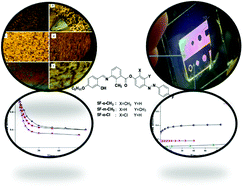
Herein, we report selectively designed, synthesized and characterized bent-core molecules bearing methyl and chloro substituents at the lateral positions on the phenyl rings of the azobenzene wings. Preliminary structural characterizations were carried out using 1H NMR, IR and mass spectroscopy. The mesomorphic and photoswitching properties were studied by using polarizing optical microscopy (POM), differential scanning calorimetry (DSC) and UV-Vis spectroscopy. All of the bent-core mesogens exhibit liquid crystalline phases and excellent photo-switching behavior. When they were irradiated with a light of 365 nm wavelength, photo-saturation occurred in around 30 s and the system took a longer time to reach its original state during thermal back-relaxation. The photoisomerization of bent-core azo compounds is very crucial for creating optical storage devices with longer thermal back-relaxation times. The devices fabricated from these materials are excellent candidates for optical image storage applications. This study provides strong evidence for the structure–property relationship, where one can tune the structures according to their behavior.


 Please wait while we load your content...
Please wait while we load your content...