Facile synthesis of SnO2@carbon nanocomposites for lithium-ion batteries†
Abstract
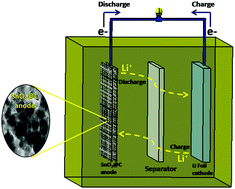
SnO2@C nanocomposites were synthesized using a citrate gel method in one step. Carbon has been used to increase the conductivity of pristine SnO2. The SnO2@C nanocomposites show nanoparticles sized in the range of 30–50 nm. A structural study by XRD shows the tetragonal rutile structure of SnO2. Furthermore, X-ray photoelectron spectroscopy (XPS) and Raman spectroscopy confirm the existence of SnO2 along with carbon. The electrochemical performance was investigated by cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS) and galvanostatic charge–discharge measurements. The nanocomposite has been used as an anode material for lithium-ion batteries. Pristine SnO2 exhibited a discharge capacity of 1854 mA h g−1 and a corresponding charge capacity of 1038 mA h g−1 for the first cycle. The SnO2@C nanocomposite showed a discharge capacity of 2574 mA h g−1 and charge capacity of 1481 mA h g−1 for the first cycle. These results clearly show the effect of carbon incorporation in SnO2. Furthermore, the nanocomposite shows a discharge capacity as high as 725 mA h g−1 after 200 cycles at a current density of 50 mA g−1. Thus, the obtained capacity for the nanocomposites is much higher as compared to that for pristine SnO2, i.e., 119 mA h g−1 at 50 mA g−1 after 200 cycles. The higher capacity in the nanocomposites is due to the fast transport of electrons and easy transport of lithium ions due to the carbon in the nanocomposites. The SnO2@C nanocomposite anode exhibits superior cycling stability and rate capability due to its stable electrode structure.


 Please wait while we load your content...
Please wait while we load your content...