Coordination control of a semicarbazide Schiff base ligand for spontaneous aggregation of a Ni2Ln2 cubane family: influence of ligand arms and carboxylate bridges on the organization of the magnetic core†
Abstract
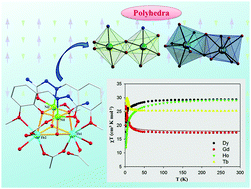
A family of tetranuclear Ni-4f coordination aggregates (CAs) from the support of a carbazido ligand is described. The option of altering the 4f ions during synthesis within the family, without altering the core topology, gave the four members of the family. Tetranuclear (2 : 2) heterometallic compounds [NiII2LnIII2(μ3-L)2(μ3-OH)2(μ-OAc)3(AcO)(H2O)3]Cl2·4H2O (Ln = Gd, 1; Tb, 2; Dy, 3; Ho, 4) have been assembled through the utilization of all the coordination sites of the anionic form of 1-(2-hydroxy-3-methoxybenzylidene)semicarbazide (HL) and bridging supports from HO− and AcO− ions. The four metal ion centers of two different types, are held together by two μ-L−, two μ3-HO− and three μ1,3-AcO− bridges to self-assemble into Ni2Ln2O4 hetero-cubane structures. In each cube, LnIII ions are linked by two hydroxido and one phenoxido bridge and each NiII ion is linked by two phenoxido and one hydroxido bridge, so that each hydroxido group in μ3-mode is linked to two LnIII and one NiII and each phenoxido ion in μ3-mode is linked to two NiII and one LnIII. Variable temperature magnetic properties have been examined to find the role of magnetic anisotropy for the single molecule magnet (SMM) behavior. All of them show large ground state magnetic moment values and absence of out-of-phase ac susceptibility signals. The absence of SMM behavior is ascribed to the lack of axial magnetic anisotropy.


 Please wait while we load your content...
Please wait while we load your content...