A molecularly engineered near-infrared-light-emitting electrochemical cell (NIR-LEC)†
Abstract
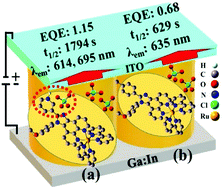
In this article, we explain how a pyridine moiety increases electroluminescence parameters including the stability of the current and luminance characteristics over time in a light-emitting electrochemical cell (LEC) compared to non-pyridine ruthenium phenanthroimidazole complexes, indicating a significant enhancement in the lifetime (t1/2) from 629 s to 1796 s in the range of 1800 s. When the ancillary ligand in the ruthenium pyridine–phenanthroimidazole family is altered from bipyridine (Bn1) to dimethyl bipyridine (Bn2), just 1% of the current density is lost in the same period. In particular, Bn1 shows two electroluminescence emission peaks at 614 nm and 695 nm, while the non-pyridine phenanthroimidazole complex indicates electroluminescence emission at 635 nm. To explore the role of the pyridine group to increase the electroluminescence properties, we propose the formation of an adduct between pyridinium (pyH+) and the counter ion ClO4− (pyH+:ClO4−) in the inner coordination sphere in Bn1, which subsequently leads to an increased number of charges and enhances the charge mobility between the two electrodes. On the basis of the obtained outstanding electroluminescence results, the main idea is to inspire readers to explore new avenues in the design of new emitters for OLEDs based on different ancillary ligand substitutions.


 Please wait while we load your content...
Please wait while we load your content...