Regioisomer effects of dibenzofuran-based bipolar host materials on yellow phosphorescent OLED device performance†
Abstract
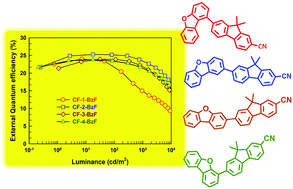
Four regioisomers were synthesized for use as bipolar host materials for phosphorescent organic light-emitting diodes (PhOLEDs) by classic cross-coupling reactions using cyanofluorene and fused dibenzofuran and were readily purified. To realize the bipolar host material, a cyano-substituted fluorene was selected as the n-type unit and dibenzofuran as the p-type unit. Yellow PhOLEDs were fabricated with iridium(III) bis(4-phenylthieno[3,2-c]pyridinato-N,C2′)acetylacetonate [PO-01] as a phosphorescent emitter. The achieved maximum current efficiency was 77.2 cd A−1 and the external quantum efficiency was 25.3% for the [PO-01]-based PhOLED; the 7-(dibenzo[b,d]furan-2-yl)-9,9-dimethyl-9H-fluorene-2-carbonitrile (CF-2-BzF) host had the best device performance. The color coordinates of yellow PhOLEDs at 1000 cd m−2 were (0.50, 0.50) (CF-1-BzF), (0.50, 0.49) (CF-2-BzF), (0.51, 0.49) (CF-3-BzF), and (0.50, 0.50) (CF-4-BzF).


 Please wait while we load your content...
Please wait while we load your content...