The pathways and domain specificity of Cu(i) binding to human metallothionein 1A†
Abstract
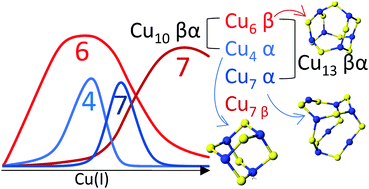
Copper is an essential element, but as a result of numerous adverse reactions, it is also a cellular toxin. Nature protects itself from these toxic reactions by binding cuprous copper to chaperones and other metalloproteins. Metallothionein has been proposed as a storage location for Cu(I) and potentially as the donor of Cu(I) to copper-dependent enzymes. We report that the addition of Cu(I) to apo recombinant human metallothionein 1a cooperatively forms a sequential series of Cu(I)–cysteinyl thiolate complexes that have specific Cu(I) : MT stoichiometries of 6 : 1, 10 : 1, and finally 13 : 1. The individual domain Cu : SCys stoichiometries were determined as Cu6S9 (for 6 : 1), Cu6S9 + Cu4S6 (for 10 : 1), and Cu6S9 + Cu7S9 (for 13 : 1) based on the number of modified free cysteines not involved in Cu(I) binding. The stoichiometries are associated with Cu–SCys cluster formation involving bridging thiols in the manner similar to the clusters formed with Cd(II) and Zn(II). The locations of these clustered species within the 20 cysteine full protein were determined from the unique speciation profiles of Cu(I) binding to the β and α domain fragments of recombinant human metallothionein 1a with 9 and 11 cysteines, respectively. Competition reactions using these domain fragments challenged Cu(I) metallation of the βα protein, allowing the sequence of cluster formation in the full protein to be determined. Relative binding constants for each Cu(I) bound are reported. The emission spectra of the Cu4S6, Cu6S9, and Cu7S9 clusters have unique λmax and phosphorescent lifetime properties. These phosphorescent data provide unambiguous supporting evidence for the presence of solvent shielded clusters reported concurrently by ESI-MS. Simulated emission spectra based on the cluster specific emission profiles matched the experimental spectra and are used to confirm that the relative concentrations seen by ESI-MS are representative of the solution. Our results suggest that the availability of a series of sequential Cu(I)–thiolate clusters provides flexibility as a means of protecting the cell from toxicity while still allowing for homeostatic control of the total copper content in the cell. This mechanism provides a dynamic and reactive method of reducing the cellular free copper concentrations.


 Please wait while we load your content...
Please wait while we load your content...