Extracellular recording of direct synaptic signals with a CMOS-nanoelectrode array†
Abstract
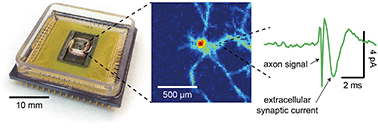
The synaptic connections between neurons are traditionally determined by correlating the action potentials (APs) of a pre-synaptic neuron and small-amplitude subthreshold potentials of a post-synaptic neuron using invasive intracellular techniques, such as patch clamping. Extracellular recording by a microelectrode array can non-invasively monitor network activities of a large number of neurons, but its reduced sensitivity usually prevents direct measurements of synaptic signals. Here, we demonstrate that a newly developed complementary metal-oxide-semiconductor (CMOS) nanoelectrode array (CNEA) is capable of extracellularly determining direct synaptic connections in dense, multi-layer cultures of dissociated rat neurons. We spatiotemporally correlate action potential signals of hundreds of active neurons, detect small (∼1 pA after averaging) extracellular synaptic signals at the region where pre-synaptic axons and post-synaptic dendrites/somas overlap, and use those signals to map synaptic connections. We use controlled stimulation to assess stimulation-dependent synaptic strengths and to titrate a synaptic blocker (CNQX: IC50 ∼ 1 μM). The new capabilities demonstrated here significantly enhance the utilities of CNEAs in connectome mapping and drug screening applications.


 Please wait while we load your content...
Please wait while we load your content...