On-chip hydrogel arrays individually encapsulating acoustic formed multicellular aggregates for high throughput drug testing†
Abstract
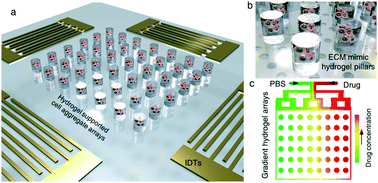
Multicellular aggregates in three-dimensional (3D) environments provide novel solid tumor models that can provide insight into in vivo drug resistance. Such models are therefore essential for developing new drugs and preventing the failure of clinical treatments. However, high-throughput cell cluster assembly and fabricating individual 3D environments that mimic the extracellular matrix (ECM) remain significant challenges. To rapidly produce mini 3D multicellular aggregate units, acoustic force assembly combined with ECM mimic hydrogel array encapsulation is developed and then integrated into a diffusion-based microfluidic device for high-throughput drug testing. The active acoustic force gathers human mononuclear leukemia cells (THP-1) into hundreds of multicellular clusters with a controllable size. Instead of continuous bulk materials, photosensitive gelatin methacryloyl (GelMA) hydrogel pillar arrays containing cell clusters at drug concentration gradients are obtained through selective area exposure. Ten azelaic acid (AZA) concentration gradient series are applied to 100 units to simultaneously test the multicellular cluster drug resistance to multiple drug conditions. Real-time green fluorescent protein (GFP) fluorescence is analyzed to monitor cell viability. The results show that cell aggregate activity is inversely related to the drug concentration in the hydrogel pillars, and shows lower sensitivity to drug toxicity than the activity of monolayer cultured cells. The 3D multicellular arrays provide numerous in vitro tumor models and can be directly used for downstream drug testing. This technology inherits the advantages of acoustic assembly, while being more flexible, practical, and high-throughput, and shows significant potential for use in further tumor related research and clinical practice.


 Please wait while we load your content...
Please wait while we load your content...