A non-phosgene process for bioderived polycarbonate with high molecular weight and advanced property profile synthesized using amino acid ionic liquids as catalysts†
Abstract
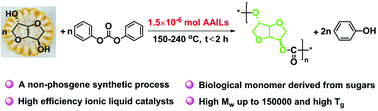
The conversion of biomass and carbon dioxide to plastics is one of the key solutions to reduce the greenhouse effect and alleviate the petroleum resource depletion. However, there is still a lack of bioderived polymers with high molecular weights and excellent performance and their corresponding green synthesis processes, which limits the potential of bioderived polymers to replace petroleum-based polymers. In this study, an eco-friendly synthetic process for bioderived polycarbonate, catalyzed by amino acid ionic liquids, was developed by utilizing isosorbide (ISO) and diphenyl carbonate (DPC) as reactants, derived from a renewable resource and carbon dioxide, respectively. By using 1-ethyl-3-methylimidazole lysine ([Emim][Lys]) as a catalyst, poly(isosorbide carbonate) (PIC) with the weight average molecular weight of 150 000, the highest reported so far to the best of our knowledge, was synthesized, and the Tg of PIC was up to 174 °C. The reaction mechanism was investigated using nuclear magnetic resonance (NMR) spectroscopy, matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-ToF MS), and density functional theory (DFT) calculation. The remarkable catalytic performance was attributed to the fact that [Emim][Lys] could effectively activate the hydroxyl group of ISO and carbonyl group of DPC, and inhibit the formation of cyclic intermediates. Moreover, to overcome the brittleness of PIC, 1,4-butanediol (BD) and 1,4-cyclohexanedimethanol (CHDM) were introduced into PIC, and the copolycarbonate showed excellent Young's modulus, ultimate tensile strength, and elongation at break, which were 979 MPa, 57 MPa, and 145%, respectively, and were comparable to the commercial petroleum-based polycarbonates. Such a process provides further industrial prospects for the next generation of bioderived polycarbonate.


 Please wait while we load your content...
Please wait while we load your content...